The Displacement Reaction
Storyboard Text
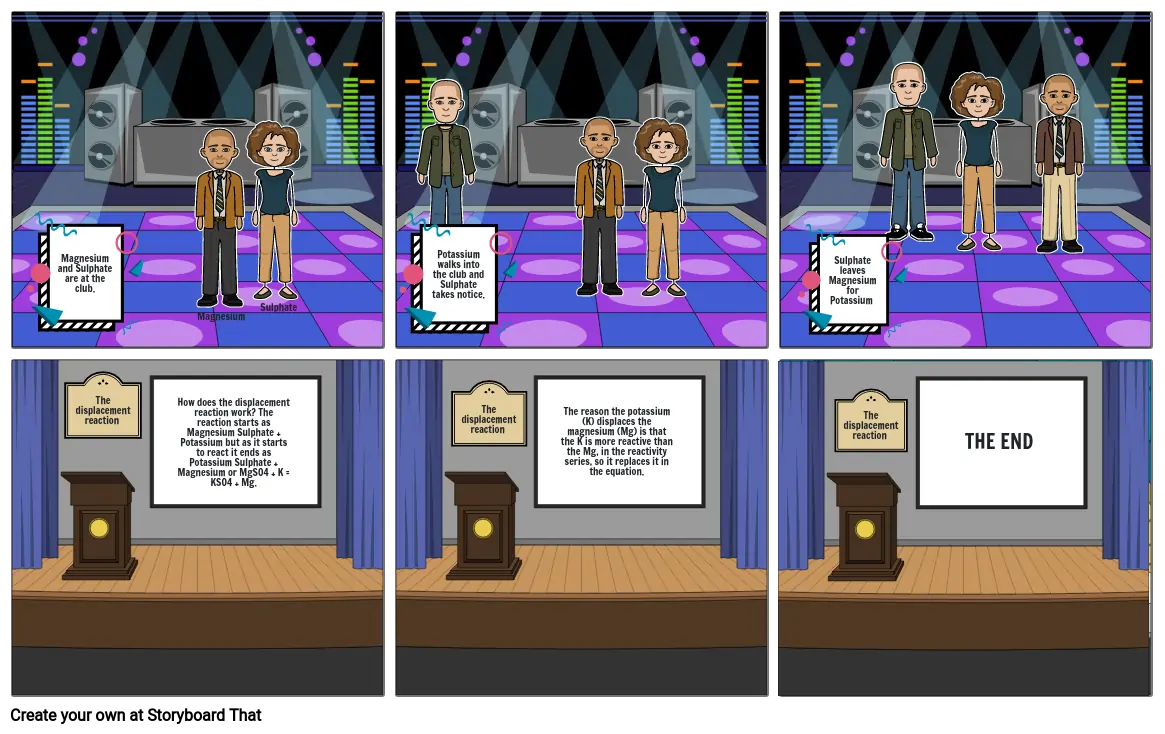
- Magnesium and Sulphate are at the club.
- Magnesium
- Sulphate
- Potassium walks into the club and Sulphate takes notice.
- Sulphate leaves Magnesium for Potassium
- The displacement reaction
- How does the displacement reaction work? The reaction starts as Magnesium Sulphate + Potassium but as it starts to react it ends as Potassium Sulphate + Magnesium or MgSO4 + K = KSO4 + Mg.
- The displacement reaction
- The reason the potassium (K) displaces the magnesium (Mg) is that the K is more reactive than the Mg, in the reactivity series, so it replaces it in the equation.
- The displacement reaction
- THE END
Over 30 Million Storyboards Created

