chem 2
Siužetinės Linijos Tekstas
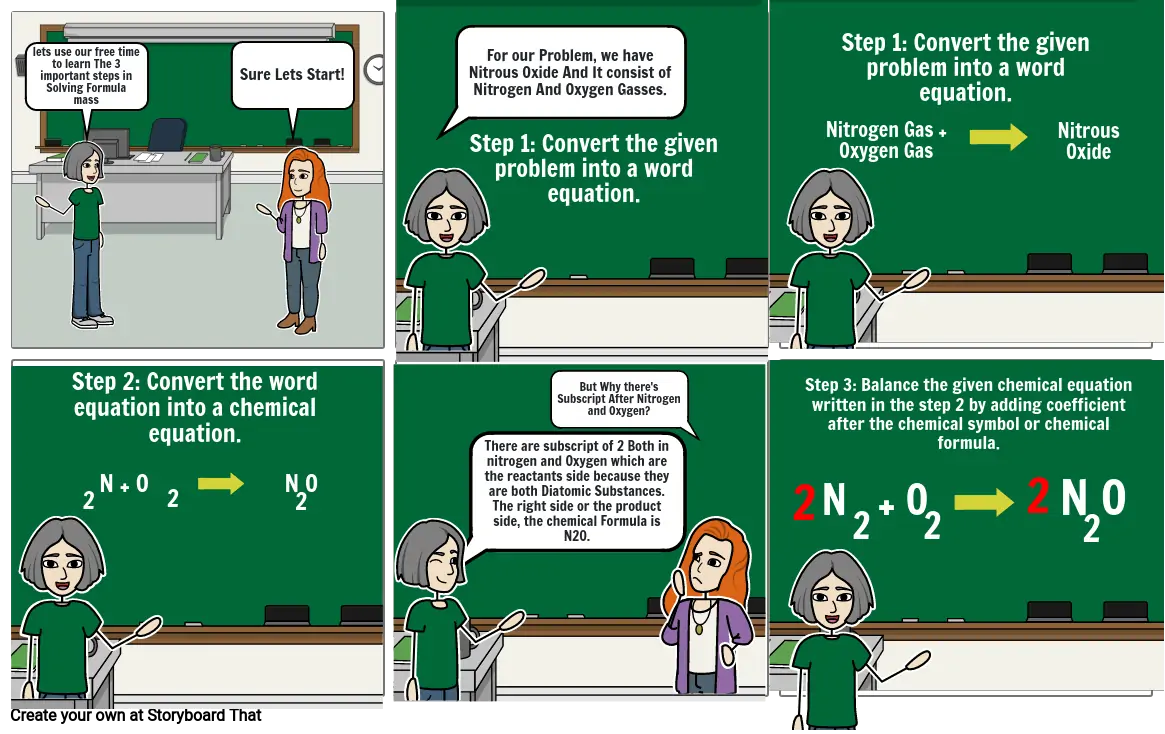
- lets use our free time to learn The 3 important steps in Solving Formula mass
- Sure Lets Start!
- For our Problem, we have Nitrous Oxide And It consist of Nitrogen And Oxygen Gasses.
- Step 1: Convert the given problem into a word equation.
-
- Nitrogen Gas + Oxygen Gas
- Step 1: Convert the given problem into a word equation.
-
-
- Nitrous Oxide
- 2
- Step 2: Convert the word equation into a chemical equation.
- N + O
-
- 2
-
- 2
- N O
- There are subscript of 2 Both in nitrogen and Oxygen which are the reactants side because they are both Diatomic Substances. The right side or the product side, the chemical Formula is N20.
-
- But Why there's Subscript After Nitrogen and Oxygen?
- 2
- N + O
- Step 3: Balance the given chemical equation written in the step 2 by adding coefficient after the chemical symbol or chemical formula.
-
- 2 2
-
-
- 2
- 2
- N O
Sukurta daugiau nei 30 milijonų siužetinių lentelių

